
What is rabies?
Rabies is one of the most dangerous viral diseases in the world, transmitted from animals to humans and almost always fatal once symptoms appear. …
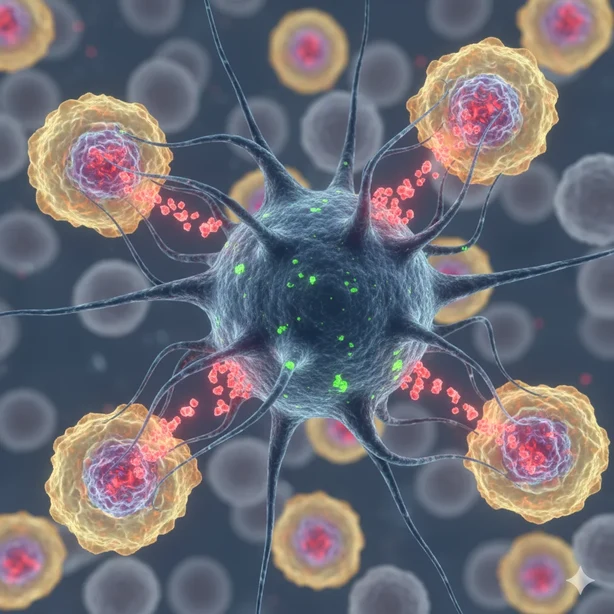
A team of researchers has successfully developed genetically engineered natural killer (NK) cells with exceptional efficiency in destroying cancer cells — achieving results faster than traditional immunotherapy methods.
The study, conducted by scientists from Harvard University and the Massachusetts Institute of Technology (MIT) in the United States, introduced a new approach in genetic engineering of natural killer cells by equipping them with Chimeric Antigen Receptors (CAR-NK). This modification significantly enhanced their ability to target and eliminate cancer cells without being attacked by the body’s immune system.
The findings were published in Nature Communications on October 8, 2025, and reported by the science platform EurekAlert.
This new method could pave the way for developing pre-prepared cancer therapies that can be administered immediately after diagnosis — unlike previous CAR-based treatments for T cells or NK cells, which required several weeks of preparation in the laboratory before use.
Genetically engineered cell therapies face numerous challenges, the most notable being the long production time needed to generate sufficient cells. In addition, cells derived from a patient’s blood are often less effective than those from healthy individuals. Using donor cells also risks rejection by the patient’s immune system, which recognizes them as foreign and destroys them before they can eliminate cancer cells.
Natural killer cells are a vital component of the human immune system, responsible for identifying and destroying cancerous or virus-infected cells. They perform this task by releasing a protein called perforin, which creates pores in target cells and triggers their death in a process known as granule exocytosis.
To produce CAR-modified NK cells, doctors first collect a blood sample from the patient, isolate the NK cells, and genetically engineer them to express receptors that recognize specific proteins on the surface of cancer cells — making them more effective in targeting and destroying tumors.
However, these modified NK cells require several weeks of expansion in the laboratory before they are ready for clinical use. The technique remains in the experimental stage, though a similar approach has already been approved for CAR-T cell therapy to treat certain blood cancers such as leukemia and lymphoma.
In this study, scientists overcame the issue of immune rejection by silencing the genes responsible for producing the HLA class I protein on the surface of donor-derived NK cells.
They also introduced additional genes to enhance cancer-fighting efficiency and prevent immune detection, combining all these genetic changes into a single DNA construct to simplify the modification process.
The researchers tested the new technique on mice with immune systems similar to humans and injected them with lymphoma cells. The genetically modified CAR-NK cells lacking HLA class I proteins successfully eradicated the cancer cells. In contrast, unmodified or partially modified NK cells were unable to survive immune attack, leading to further tumor growth.
The research team hopes to advance this technique to clinical trials in humans, potentially replacing current low-efficacy treatments for lymphoid and other cancers. The breakthrough may also open the door to treating autoimmune diseases such as lupus in the future.

Rabies is one of the most dangerous viral diseases in the world, transmitted from animals to humans and almost always fatal once symptoms appear. …

Nestlé has announced a precautionary recall of selected infant formula products in several markets after detecting a potential contamination risk that could pose health …

A recent study has raised serious concerns about the impact of private equity ownership on U.S. hospitals, revealing a significant rise in emergency department …
In a groundbreaking leap for regenerative medicine, scientists have developed a tiny bioprinter—no larger than a pill—that can enter the digestive system and print …